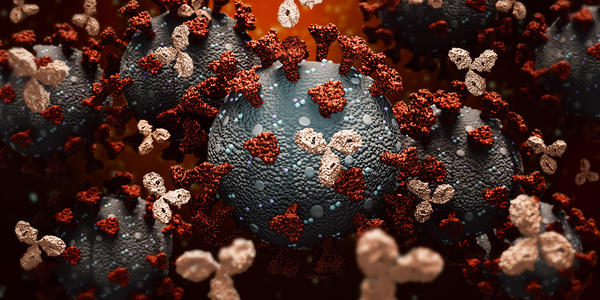
Monoclonal Antibody Therapy Was Effective for Earlier Variants of COVID-19
In January 2023, the FDA pulled the emergency use authorization for the monoclonal antibody therapy EVUSHELD because it no longer protects against the newer, dominant strains of COVID-19 in the United States. In late 2022, the FDA also pulled the authorization for bebtelovimab for the same reason.

According to the CDC, EVUSHELD is unlikely to work against Omicron BQ.1, BQ.1.1, XBB and XBB.1.5 subvariants. Several other monoclonal antibody therapies used earlier in the pandemic are also no longer available because they were not effective when the virus evolved. For patients with COVID-19, antiviral medications may be treatment options.
- Antiviral oral medications (Paxlovid™ and molnupiravir) are widely available for treating mild-to-moderate COVID-19. These medications are given to patients who meet eligibility requirements outlined by the FDA.
- Remdesivir (Velkury®) is available as a three-day (once each day) intravenous infusion therapy for patients who meet eligibility requirements outlined by the FDA.
COVID-19 vaccination remains the most effective way to prevent infection, severe illness, hospitalization and death. Wearing a well-fitting face mask, limiting time in crowded indoor spaces and getting tested if you have been exposed to the virus are additional important prevention measures.
Note: the information in the article below was accurate at the time of publication through 2022.
Antibodies are proteins created by certain cells in your immune system to recognize and destroy harmful microorganisms, such as bacteria or viruses. After fighting off the infection, the cells are able to remember the invaders, quickly recognize them and produce protective antibodies again. In November 2020, the FDA authorized emergency use of monoclonal antibodies to treat eligible COVID-19 patients. The treatment is an infusion and is available at some Froedtert & the Medical College of Wisconsin locations.
What Are Monoclonal Antibodies?
Monoclonal antibodies are man-made versions of the antibody proteins made by the human body. Throughout the COVID-19 pandemic, clinicians and researchers have been working on a coronavirus cure. One effort has been to determine which antibodies can fight the COVID-19 virus, isolate them, replicate them in a lab setting and then mass produce them to use as treatment.
Is Monoclonal Antibody Therapy Effective Against COVID-19?
In January 2023, the FDA pulled the emergency use authorization for the monoclonal antibody therapy EVUSHELD because it doesn’t protect against the newer, dominant strains of COVID-19 in the United States. In late 2022, the FDA also pulled the authorization for bebtelovimab for the same reason.
In 2020, clinical trials with early variants of COVID-19 showed that some monoclonal antibody cocktails, such as bamlanivimab, with etesevimab, casirivimab and imdemivab, or sotrivimab used alone, prevented hospitalizations and emergency room visits in people with mild to moderate symptoms of COVID-19. The monoclonal antibodies stopped the disease from progressing by quickly reducing the quantity of virus in the infected person’s blood. The existing data prompted the FDA to issue emergency use authorizations to drug makers Eli Lilly and Company, Regeneron Pharmaceuticals and GlaxoSmithKline, respectively.
In early 2022, the FDA restricted the use of certain monoclonal antibody treatments because new data showed they were not effective against the omicron variant, which had become the dominant variant of COVID-19. Omicron and its highly contagious subvariants are resistant to many monoclonal antibody treatments. The only monoclonal antibody infusion that is effective against the omicron variant is bebtelovimab. Oral antiviral medications are another option.
The medications Paxlovid and molnupiravir are effective against the omicron variant and are widely available to people who meet the FDA’s eligibility criteria.
How Does Monoclonal Antibody Therapy Work?
Monoclonal antibody therapy is a single infusion given in an outpatient setting. The treatment works by providing the body with a large quantity of ready-made antibodies that attach to the virus particle that causes COVID-19 and prevents the virus from attaching to and entering cells in the body. The infusion should be given as soon as possible after a positive COVID-19 test and within 10 days of the onset of COVID symptoms.
“COVID-19 has two phases,” said Njeri Wainaina, MD, infectious disease specialist with the Froedtert & MCW health network. “The first phase is caused directly by the virus, and the second phase, which is more severe, is due to inflammation that is driven by the body’s immune response. The second phase can be avoided by treating the first phase. If we can block the virus from entering cells and reduce the extent of infection, we can effectively prevent severe COVID-19.”
Who Is Eligible for Monoclonal Antibody Therapy?
In January 2023, the FDA pulled the emergency use authorization for the monoclonal antibody therapy EVUSHELD because it doesn’t protect against the newer, dominant strains of COVID-19 in the United States. In late 2022, the FDA also pulled the authorization for bebtelovimab for the same reason.
Monoclonal Antibody Infusions
According to the FDA, bebtelovimab is only available to patients who are at high risk for progression to severe COVID-19, have tested positive for COVID-19, have mild to moderate COVID-19 symptoms and for whom alternative COVID-19 treatments are not accessible or clinically appropriate. Adults who meet at least one of the following criteria are defined as high risk by the FDA:
- Are 65 years old or older
- Have a body mass index (BMI) over 25
- Have chronic kidney disease
- Have diabetes
- Are pregnant
- Have immunosuppressive disease
- Are currently receiving immunosuppressive treatment (for example, cancer patients or transplant patients)
- Have cardiovascular disease or hypertension
- Have chronic lung disease (for example, chronic obstructive pulmonary disease or COPD, moderate to severe asthma, interstitial lung disease, cystic fibrosis or pulmonary hypertension)
- Have sickle cell disease
- Have a neurodevelopmental disorder like cerebral palsy, or other conditions that confer medical complexity (for example, genetic or metabolic syndromes and severe congenital anomalies)
- Have a medical-related technological dependence (for example, tracheostomy, gastrostomy or positive pressure ventilation not related to COVID-19)
People who are currently hospitalized due to COVID-19 or who require supplemental oxygen due to COVID-19 are not eligible for monoclonal antibody therapy.
All other previously available monoclonal antibody infusions for COVID-19 are restricted by the FDA until further notice because they are not effective against the omicron variant. Based on CDC data, the majority of COVID-19 infections in early 2022 were due to the omicron variant, so treatment with bebtelovimab is currently the most effective monoclonal antibody therapy.
Monoclonal Antibody Injection
In January 2023, the FDA pulled the emergency use authorization for the monoclonal antibody therapy EVUSHELD because it doesn’t protect against the newer, dominant strains of COVID-19 in the United States. In late 2022, the FDA also pulled the authorization for bebtelovimab for the same reason.
For people who are not infected with COVID-19 and who have not recently been exposed to COVID-19, a new monoclonal antibody injection, EVUSHELD™, is available. EVUSHELD is a combination of two long-acting antibodies, tixagevimab with cilgavimab. It offers protection against COVID-19, which is why it is a called a COVID-19 pre-exposure prophylaxis monoclonal antibody. It is given by two injections, one after another. Eligibility for EVUSHELD is determined based on the FDA’s emergency use authorization.
EVUSHELD is not a substitute for the COVID-19 vaccine, but it can help prevent COVID-19 for people with compromised immune systems, people who may not receive adequate immunity from COVID-19 vaccines or people with severe allergies to COVID-19 vaccines. The Froedtert & MCW health network is receiving limited supply of this medication from the state and cannot offer it to all patients. The hope is to expand access as soon as supply increases.
How Can I Get Monoclonal Antibodies for COVID-19?
In January 2023, the FDA pulled the emergency use authorization for the monoclonal antibody therapy EVUSHELD because it doesn’t protect against the newer, dominant strains of COVID-19 in the United States. In late 2022, the FDA also pulled the authorization for bebtelovimab for the same reason.
In 2021, the Froedtert & MCW health network started offering monoclonal antibody therapy at two of its infusion clinics. As the dominant variant of COVID-19 changed from delta to omicron, bamlanivimab/etesevimab, casirivimab/imdevimab and sotrovimab monoclonal antibody infusions were suspended, as they do not protect against omicron or its subvariants. Monoclonal antibody infusions of bebtelovimab — which is effective against the omicron variant — are available, but supplies are limited.
The U.S. Department of Health and Human Services is responsible for overseeing the distribution of monoclonal antibody supplies to individual states. Quantities allocated to individual states are determined based on each state’s percentage of confirmed COVID-19 cases and weekly COVID-19 hospitalization rates. In Wisconsin, a subcommittee of the State Disaster Medical Advisory Committee, a part of the Wisconsin Department of Health Services, was responsible for determining how these medications would be equitably distributed to local health systems. Local health systems place orders to ensure they have the supplies needed to provide treatment to their patients.
The Froedtert & MCW health network created a centralized process to identify and assess risk level for patients who recently tested positive for COVID-19 and were eligible for monoclonal antibodies.
“We used the FDA’s criteria to determine which patients we would be able to offer the treatment to,” Dr. Wainaina said. “Then, we had to find a way to identify these people in a timely manner.”
Mark Lodes, MD, a Froedtert & MCW internal medicine and pediatric physician and vice president and chief medical officer for Froedtert & MCW Population Health and Medical Education, worked with Dr. Wainaina to design the identification process.
- Patients who have COVID-19 symptoms and test positive for COVID-19 through a Froedtert & MCW lab will be referred automatically for monoclonal antibody therapy if they are eligible.
- Patients who have symptoms and test positive through another lab but have a Froedtert & MCW provider should contact that provider for referral for monoclonal antibody therapy if it is right for them.
- People who don’t have a provider with our network but want to be considered for monoclonal antibody therapy can complete an E-Visit to get tested through our lab. If positive and eligible, they may be offered treatment.
“We use our internal databases to help us pinpoint eligible patients who get a positive COVID-19 test result at Froedtert & MCW test sites,” Dr. Lodes said. “When a person schedules a test, we gather the information we need on risk factors based on the authorizations from the FDA that could qualify them for monoclonal antibody therapy. If their test is positive, the patient’s case is pulled into an internal registry. Our system generates a report for us each day of patients who have had positive COVID-19 tests, including which criteria they meet for the treatment.”
If, after a positive COVID-19 test, a patient is eligible for monoclonal antibody therapy, a nurse from the Froedtert & MCW virtual care team will proactively reach out to them to offer the treatment. Initial outreach takes place electronically through MyChart. This is followed by a phone call to schedule treatment. Patients who do not use MyChart will receive a phone call. By August 2021, the Froedtert & MCW health network had treated more than 1,100 patients for COVID-19 with monoclonal antibodies.
“We have developed a fast and efficient way to pinpoint which of our patients can benefit from this treatment,” Dr. Lodes said. “We are very encouraged by the results. Our data is in line with the clinical trials that support the FDA authorizations. We are seeing an extremely low percentage of patients who have been treated with monoclonal antibody therapy needing to be hospitalized for further COVID-19 treatment.”
The health network’s patient identification process for monoclonal antibody therapy was a collaborative effort among Froedtert & MCW leadership and several teams, including infectious disease, population health, information technology, pharmacy, infusion clinic staff and the virtual care team through Inception Health, the hub for the health network’s digital health services.
“Everyone put their best foot forward,” Dr. Wainaina said. “Figuring this out together felt like being a part of a winning Olympic team.”
Can I Get the COVID-19 Vaccine After Monoclonal Antibody Therapy?
The CDC no longer recommends a waiting period of 90 days to receive the vaccine. COVID-19 vaccination can be given at any interval following receipt of passive antibody therapy (convalescent plasma or monoclonal antibodies) at least until recovery from the acute illness (if symptoms were present) and criteria to discontinue isolation have been met.
Learn more about COVID-19 testing and care options.



I have a DM patient who meets criteria- how do I refer in?
Thank you for inquiring. We have followed up with you via e-mail with more information.
I am a local clinician, can I get information on how to refer and contact the Monoclonal Ab Clinic?
Thank you for inquiring. We have followed up with you via e-mail with more information.
I'm interested in learning more about this program, thank you!
Hi Sarah - Thank you for your inquiry. We recommend that you reach out to your primary care provider so they can assess your condition. If they determine that monoclonal antibody therapy is appropriate for your care, they can refer you to our program.
How do I refer patients?
Thank you for inquiring. We have followed up with you via e-mail with more information.
I have a patient who meets criteria. I am at Aurora but would have to send her to ER. Can I refer her to your clinic?
Thank you for your inquiry. Yes, you can refer a patient. We have followed up with you via e-mail with more information.
I was told when I called the Froedtert COVID hotline today, looking for an infusion for a family member who meets criteria and was tested by Froedtert, that only 6-10 infusions are being offered per day for the greater Milwaukee area. And even if you’re eligible, if you aren’t in the top 6-10 of the current 89 or so eligible/day you will not get an infusion. And even though you should be eligible for the first 10 days, names that don’t make the list, are not carried over. Is this accurate information?
Hi Sara - Thank you for reaching out. If you are interested in learning more about mAb therapy for you or your family member, or wish to pursue therapy, please contact the primary care provider of the patient to discuss further. If the primary care provider believes you are a candidate for mAb therapy, they can make a referral by calling 263-532-3390. Thanks, Taylor
i am a local primary care doc, how do I refer patients.
Thank you for your inquiry. We have followed up with you via e-mail with more information. - Taylor
Hi my Dr is Aurora Dr and I have a Dr at Mayo Clinic. Mayos wanted me to receive this treatment if I am exposed due to Medical history and immune suppressed if exposed to a positive person or infected. How can I tell my Dr to refer me from Aurora? Mayos told me to go to Primary but she doesn’t know how to get treatment for me. Thank you
Hi Renee - Thank you for reaching out. If your provider believes you are a candidate for mAb therapy, they can make a referral by calling 262-532-3390. Thanks, Taylor
In Florida a person can walk into a monoclonal facility with covid and get treatment free of charge. Does Wisconsin have any type of facility or do patients have to have a referral? Which for some us, is hard enough to get!
Thank you for your inquiry. We recommend that individuals who are interested in receiving monoclonal antibody therapy reach out to their primary care providers so they can assess the patient’s condition. If they determine that monoclonal antibody therapy is appropriate for care, they can refer patients to our program.
How long will the Monoclonal Antibody treatment be available at Froedtert? Does Froedtert have to put in requests to the Federal government to obtain more of this type of treatment every month?
Hi Lori - We plan to continue offering COVID-19 monoclonal antibody therapy for the foreseeable future. There will likely be ongoing developments in this and other antiviral therapies, so our offering will evolve to reflect the latest advancements available. Monoclonal antibodies are approved under emergency use authorization, so they are provided to the government, and then the government distributes them to hospitals and health systems. Currently, the federal government provides this resource to each state. We provide the state of Wisconsin with information about our weekly usage of the medication, and they supply us with periodic shipments to maintain our inventory.
Do you make treatment exceptions for people under 55 with no comorbidities but are experiencing severe symptoms (within 10 days of infection)?
Hi John - Patients need to have one or more of the factors/conditions that define them as high risk in order to eligible under the FDA’s Emergency Use Authorization. Those factors are listed in the "Who Is Eligible for Monoclonal Antibody Therapy?" section of this article. If patients do not meet at least one of these factors, they are not eligible for infusion. Patients experiencing severe symptoms should seek medical attention.
Do you give monoclonal antibodies for covid in Kenosha at all?
Hi Sue - We offer monoclonal antibody therapy to appropriate patients throughout the area, but our program does not have a specific location in Kenosha. We suggest that you consult with your primary care provider to find out if this treatment is right for you and where best to receive it.
What types of side effects are people having with vaccinations after receiving monoclonal antibody therapy?
Thank you for your question. The CDC recommends that individuals wait 90 days before receiving a COVID-19 vaccine after receiving monoclonal antibodies for the treatment of COVID-19 infection. This is because the antibody infusion might blunt the native immune response to the vaccine (might make the vaccine less effective) in the 90 days following infusion.
I live in a border community. Is your database based on residence of the individual or on where the test occurred?
Our registry is built off of positive COVID tests done within our health system.
Can a fully vaccinated (2 Pfizer doses plus booster) who tests positive for COVID, has mild symptoms and meets criteria (BMI greater than 25) receive the monoclonal antibody infusion treatment?
Hi Allie - Patients need to have one or more of the factors/conditions that define them as high risk in order to eligible under the FDA’s Emergency Use Authorization. Those factors are listed in the "Who Is Eligible for Monoclonal Antibody Therapy?" section of this article. We recommend that individuals who are interested in receiving monoclonal antibody therapy reach out to their primary care providers so they can assess the patient’s condition. If they determine that monoclonal antibody therapy is appropriate for care, they can refer patients to our program.
Where can i go to get antibodies to fight Covid 19?
Hi Judy, We recommend that you discuss this with your primary care provider, who can refer you to our program if appropriate.
Can someone who has diabetes, hypertension, bmi over 25, and has yearly bronchitis have monoclonal antibodies and is 12 days past the 1st symptom? Do you have to be a Froedtert patient?
Hi Jeannine - Patients need to have one or more of the factors/conditions that define them as high risk in order to eligible under the FDA’s Emergency Use Authorization. Those factors are listed in the "Who Is Eligible for Monoclonal Antibody Therapy?" section of this article.
Patients who have COVID-19 symptoms and test positive for COVID-19 through a Froedtert & MCW lab will be referred automatically for monoclonal antibody therapy if they are eligible. Patients who have symptoms and test positive through another lab but have a Froedtert & MCW provider should contact that provider for referral to the monoclonal antibody program if it is right for them. People who don’t have a provider with our network but want to be considered for monoclonal antibody therapy can complete an e-visit at froedtert.com/e-visits to get tested through our lab. If positive and eligible, they may be offered treatment.
Please note that we are receiving limited supplies of these medications from the state at this time and unfortunately are unable to offer this service to all patients. Our desire is to expand access as soon as the supply increases.
Can anyone do this or do you have to be a patient at MCW??
Thanks for your question, Stephanie. We recommend that you discuss this with your primary care provider, who can refer you to our program if appropriate.
What about 2nd dose of Evusheld. 6 months after first dose?? Still @ Moreland rd clinic Froedtert
Hi Lee. Thanks for your question. We often recommend that you discuss this with your primary care provider, as everyone's situation can be different.